Roman waterpipe, made of soldered lead strips (Museum of Arles et de la Provence antique)
Historical and technical introduction of fusible alloys and fire safety fusible links
© by Jacques Jumeau 2018/08/22
The history of low temperature fusible alloys is a succession of stages, spread over two millennia, according to the successive discoveries of metals and experiments.

THE 183°C LIMIT: THE BINARY ALLOYS OF LEAD AND TIN
The earliest known piece made of lead and tin alloy seems to be an Egyptian vase found in Abydos, dated around 1400 BC.
During the Roman Empire, lead was used for the construction of water pipes. Melting at 325°C, it was easily melted into strips. As it does not self-weld, it was a mixture of lead and tin that was used to weld the strips rolled together into hoses. Although they did not have temperature measuring devices, the Romans had noticed that by adding a certain percentage of tin (melting at 235°C) imported from Cornwall to the lead, the mixture melted at a temperature less than that of lead. In his Natural History, Pliny the Elder, in the course of the first century, gave the formula for welding the lead tubes: two parts of lead for a part of tin. (Melting range of the alloy 66.7-33.3: 185-250°C).
Alloys with 4 parts of lead and one part of tin (melting range for the 80-20 alloy: 183-275°C) and 5 parts of lead and one part of tin (melting range of the alloy 83.3-16.7: 225-290°C) are then given for a temperature of 81.3.3 / 4 according to the Isaac Newton scale in 1701.
Still in the middle of the 18th century, this anomaly in the alloys always intrigued and remained unexplained “One thing that is still rather
singular; it is because any two metals mixed together are melted at a lower fire than if they were separated. ” (Dissertation on the nature and propagation of fire, by the Marquise Du Chatelet, 1744)
In the 18th century, tinsmiths used a solder with 50% lead and 50% tin (melting range 183-216°C). For tin potters it was still not enough because too close to the melting temperature of the tin. It is likely that it was the Cornish tin potters who found the binary alloy with the lowest melting point, made of 63% tin and 37% lead (3 parts of lead and 5 parts of tin). At the beginning of the 18th century this eutectic alloy melting at 183°C was commonly used for tinning copper kitchen vessels. Nowadays is still used as a welding alloy in the industry.
THE 96°C LIMIT: THE BISMUTH
It seems that the ancient Egyptians used bismuth oxide as a component of makeup and cosmetics “The White of Egypt”. In 1413, Basil Valentine recorded it for the first time in the following terms: “Antimony is the bastard of lead, just as wismulh, or marcasite, is the bastard of tin”. In a treatise of Agricola dating from the beginning of the 16th century (1529) it is described as being well known in Germany and considered as a particular metal. Others considered it a kind of lead.
Bismuth was later extensively described in Moyse Charas’s “Royal Galenic and Chymyque Royal Pharmacopoeia” in 1676, but its extraction and purification from tin or copper ores was complex.
The miners of the time considered bismuth as silver not yet completely transmuted and named its ore “Argenti tectum” (M. Hellot, Memoirs of the French Academy, 1737, p 231) In 1701, the first low-temperature ternary alloys using bismuth tin and lead were described by Isaac Newton in his article “Scalum graduum Caloris” (Philosophical Transactions, 1701, 270, P824-82) to serve as a reference point for thermometer calibrations. In this article in Latin, he described in particular an alloy consisting of 2 parts of lead (20%), 3 parts of tin (30%) and 5 parts of Bismuth (50%). This alloy is the one he considered to have the lowest melting point. He gave its temperature (graduated 34 1/2 in its scale) as being slightly higher than that of boiling water. (An alloy of this composition made with current pure metals is characterized by a liquidus temperature at 123°C and solidus at 96°C). He explored other ternary alloys of the same type, and also tin bismuth binary alloys. At that time, tin ore founders in the province of Cornwall used bismuth to make their tin shiny, hard and sonorous.

1701 DESCRIPTION OF AN ALLOY COMPRISING 2 PARTS OF LEAD, 3 PARTS OF TIN AND 5 PARTS OF BISMUTH BY ISAAC NEWTON IN “SCALUM GRADUUM CALORIS”
Studied empirically from the second half of the 18th century, the composition of these alloys varied as the development of increasingly pure metals.
In the second half of the eighteenth century, tin potters used many different types of welding, more or less secret, composed of lead, tin and bismuth (article “soudure”of the Encyclopédie, ou Dictionnaire raisonné des sciences, des arts et des métiers, 1775)
In 1753, the French scientist Claude Geoffroy The Young devoted himself to the study of Bismuth, which he described as a new metal and no longer as a semi-metal close to lead as it was previously considered. He died unfortunately before finishing his work.
During his life, the German pharmacist Valentin Rose the Elder (1736-1771), studied different compositions of Bismuth, lead, tin alloys with low melting point of variable composition, which were only posthumously published in 1772. He left his name to one of them. In 1775, the French chemist Jean d’Arcet provided the Academy of Sciences with a report of his experiments on fusible alloys of lead, bismuth and tin, which had the particularity of melting in boiling water. They differed from previous alloys whose melting points (liquidus) were always higher than 100°C and only solidification (solidus) was below the boiling water. He described a set of more than ten compositional variations that were then known as D’Arcet or Darcet’s Alloys. It was not until 1898 that the French chemist Georges Charpy revealed that there was only one eutectic point at 96°C for these ternary alloys, for a combination by weight of 52% Bismuth, 32% lead , and 16% tin. (“On the constitution of eutectic alloys, G. Charpy”). Many compositional variations close to this eutectic gave melting points approaching a few degrees, with a more or less extensive pasty zone, and could not therefore be considered as eutectic alloys.
The first application of one of these alloys melting at 98°C, consisting of three parts of tin, eight parts of bismuth and five parts of lead, were anatomical injections, and the manufacture of stereotyped printing plates.
Some of these ternary alloys of bismuth, tin and lead, took the name of their inventors:
- The Rose’s alloy (50% bismuth, 25-28% lead and 22-25% tin, with a melting point between 94°C and 98°C),
- The Newton’s alloy, with a melting point at 95°C, comprising 50% of bismuth, 31% of lead and 19% of tin (NB:This composition does not correspond to its description of 1701).
- The Lichtenberg’s alloy, melting at 92°C, contains 50% bismuth, 30% lead and 20%
- The Malotte’s metal melting at 95°C (203°F) contains 46% bismuth, 20% lead and 34%
- The Homberg’s alloy, melting at 121°C, contains 3 parts of lead, 3 of tin and 3 of
In 1802 the British Richard Trevithick and Andrew Vivian invented the first high-pressure steam engine that opened the way for locomotives, the first of which was used in February 1804. In this vehicle, a fuse plug in lead in the bottom of the boiler served as a temperature safety device, and its melting was supposed to send a jet of steam, extinguishing the hearth below. A second plug, made of lower temperature fusible alloy, and located at the top of the boiler, in contact with the steam was supposed to melt when the temperature of it became too high.
Although quickly considered unreliable and usable only as an auxiliary safety device, fusing plugs and fusible washers quickly became mandato- ry on steam engines: as of October 29, 1813, a decree of the French government forced the steam engine manufacturers, in addition to the safety valves, to apply a fusible plug on the boiler melting at a temperature below the maximum permitted temperature.
As early as 1821, it is proposed to make them mandatory also on pressure cookers of the “Papin’s pot” type (Annals of the National and Foreign Industry, or
Technological Mercury, 1821, p14).
A little after, the decree of October 28, 1823 imposed in France the use of two fusible plugs of different sizes on high pressure boilers (more than 2 kg/cm²), one at 10°C, the other at 20°C in below the maximum limit of the boiler. In 1828, the fusible alloy washers melting temperature, already used since several years on the safety valves of the steam locomotives, must melt at 20°C higher than that of the stamp of the boiler.
The 100°C alloy is then given as composed of 8 parts of Bismuth, 5 parts of lead and 3 parts of tin. (Steam Engineers’ Manual, by Janvier, 1828). In 1830, the bulletin of the laws further enacts “It will be further adapted to the upper part of each boiler, and near one of the safety valves, a metal washer fusing at the temperature of 127°C ”
Different tables were established for the realization of fusible alloys for boilers. This elaboration of fusible alloys at various temperatures did not take in account the notion of eutectics, and was fatal to this application on boilers: the most fusible part of these alloys (the eutectic) gradually melting and disappearing and leaving in the washer the surplus of metals melting at a significantly higher temperature. The mandatory use of these fusible alloys washers and plugs for the safety of steam boilers. was abandoned in the government ordinances dated 22 and 23 of May 1843.

Fusible alloys composition used in steam machines (1828, Traité des machines à vapeur et de leur application à la navigation, Thomas Tredgold)
1832 Locomotive fuse plug (b), combined with a shut-off valve from Mr. Edward Hall (Bulletin de la Société d’Encouragement pour l’Industrie Nationale)
1847 Fusible plugs on boilers of steam locomotives. Cap “e” melts and releases steam (US patent # N°5022, Alfred Stillman)
| Bismuth | Plomb | Étain | Tension de la vapeur en atmosphère | Températures correspondantes en degrés centigrades |
| Parties | Parties | Parties | Atmosphères | Degrés |
| 8 | 6,44 | 3 | 1 | 100 |
| 8 | 8 | 3.80 | 1 1/2 | 112.2 |
| 8 | 8 | 7,5 | 2 | 122 |
| 8 | 9,69 | 8 | 2 1/2 | 129 |
| 8 | 12,64 | 8 | 3 | 135 |
| 8 | 13,30 | 8 | 3 1/2 | 140,7 |
| 8 | 15 | 8 | 4 1/2 | 145,2 |
| 8 | 16 | 9 | 5 | 150 |
| 8 | 16 | 19 | 5 1/2 | 154 |
| 8 | 25,15 | 24 | 6 | 158 |
| 8 | 27,33 | 24 | 6 1/2 | 164 |
| 8 | 28,66 | 24 | 7 | 168 |
| 8 | 29,41 | 24 | 7 1/2 | 170 |
| 8 | 35,24 | 24 | 8 | 173 |
Fusible alloy for steam machines (1875 Grand dictionnaire universel, volume 15, Larousse)
However, in the middle of the 19th century, Darcet’s low-temperature fusible alloys were widely used in industry, including metal molds for electroplating, which after use only left the outer layer of copper, thus realizing hollow objects, also allowed easier bending of tubes filled with these alloys, but also a machine named of “internal combustion” supposed to replace the steam engines to pump the water, invented in 1839 by Antoine Galy-Cazalat (often taken under the name of Galli by his laudators), professor of physics at the Royal College of Versailles, in which the fusible alloy, heated, served as a movable liquid plug and whose displacement in a spiral produced a movement.
THE 72°C LIMIT: CADMIUM
In 1817 Friedrich Stromeyer was the first to produce cadmium. But it was not until more than 30 years ago that lead, tin, bismuth and cadmium quaternary alloys appeared. The addition of cadmium reduced the melting temperature from 20 to 25°C, and went down to 72°C.
The arrival of the fire detection systems between 1860 and 1890 (alarm or sprinklers) led to the development of all current fire detection fusible links.
The alloy invented and patented in the USA in 1860 by the American dentist Barnabas Wood, who was later named in his honor “Wood Alloy”, was first used in dentistry. It was then the first metal used for automatic sprinklers. It contains 50% bismuth, 27.6% lead, 13.4% tin and 10% cadmium. His discovery was widely commented on in Europe. (“On a New Highly Fusible Alloy,” Appl. Chem. Rep., 1860, 2, 313-314 and Wood’s Leichtflüssiges Metall, “Dingler’s Polytech, J., 1860, 158, 271-272.). It melted at 70-72°C (158-160°F) and was then adopted as the operating temperature for sprinkler plugs in the United States and most other countries. This alloy was long time given to the USA as a 155°F alloy (68°C).
In the same year, the Berlin chemist Friedrich Julius Alexander Lipowitz, referring to the discovery of Wood, invented a close alloy: with 50% bismuth, 27% lead, 13% tin, 10% cadmium, very ductile, melting between 70-74°C. The melting point of the Lipowitz alloy, which it says is at 60°C, is only 70°C, but the confusion may be due to the fact that it also tried to introduce mercury into this alloy, which lowered its melting point to 60°C. (Polytechnisches Journal, 158, 376, 1860).
A few years later, Frederick Guthrie, in the articles he wrote in the Philosophical Magazine between 1875 and 1884 on eutectic alloys, described among others the alloy at 47.4% of bismuth, 19.4% of lead, 20% tin and 13.2% cadmium. He created in 1875, on a Greek root, the term “eutectic”. (N.B.: The compositions and melting temperatures of these various alloys are clearly described in the “Encyclopedie Chimique” of Fremy, published in 1888, and may vary according to the sources, the names of the inventors being often associated with several alloy compositions).
The first fusible links appeared around 1882, and were used to command the opening of valves sending water into the fire pipes. Very quickly, the creep under permanent stress and temperature of the fusible alloys showed the possible load limits, and as early as1883 appeared the de-multiplied mechanisms.
Around 1880, the development of electrical appliances and electrical distribution networks brought out a new family of devices using fusible alloys: the fire detection electric switch, in which the melting of the alloy closed an electric circuit of alarm, either powered by batteries is powered by the network.
It was not until 1912 that the melting temperature of the eutectic alloy made of lead cadmium, tin, bismuth was confirmed at 70°C as being as low as possible with these components, but the habit was taken of naming it alloy to 72°C. (Parravano and Sirovich, Quaternary Alloys of Lead, Cadmium, Bismuth and Tin, Gazz. Chim. Ital., 42, I, p. 630; 1912)
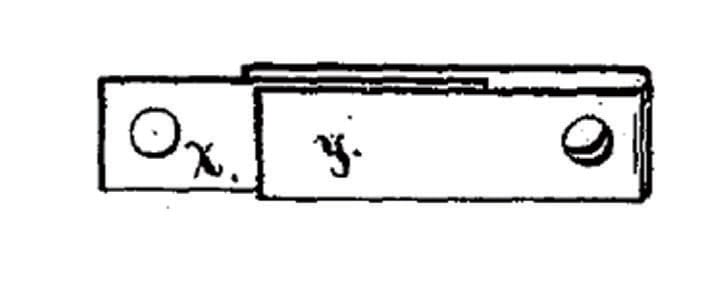
1882 Simple Fusible link used on a cable, invented by Frederick Grinnel (US patent #269.199)
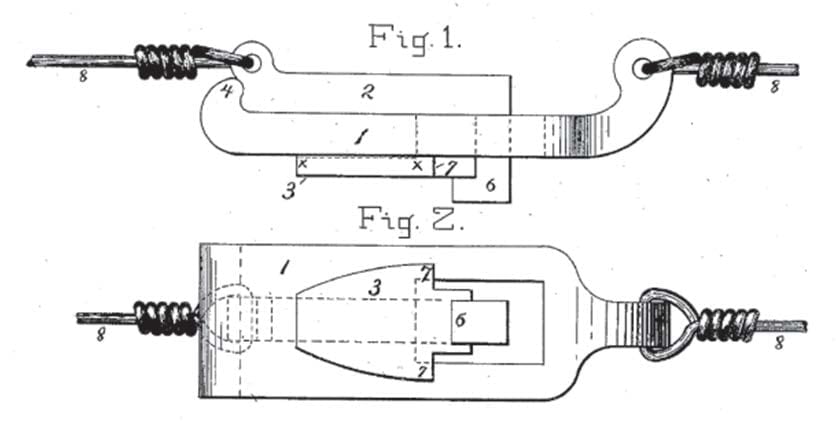
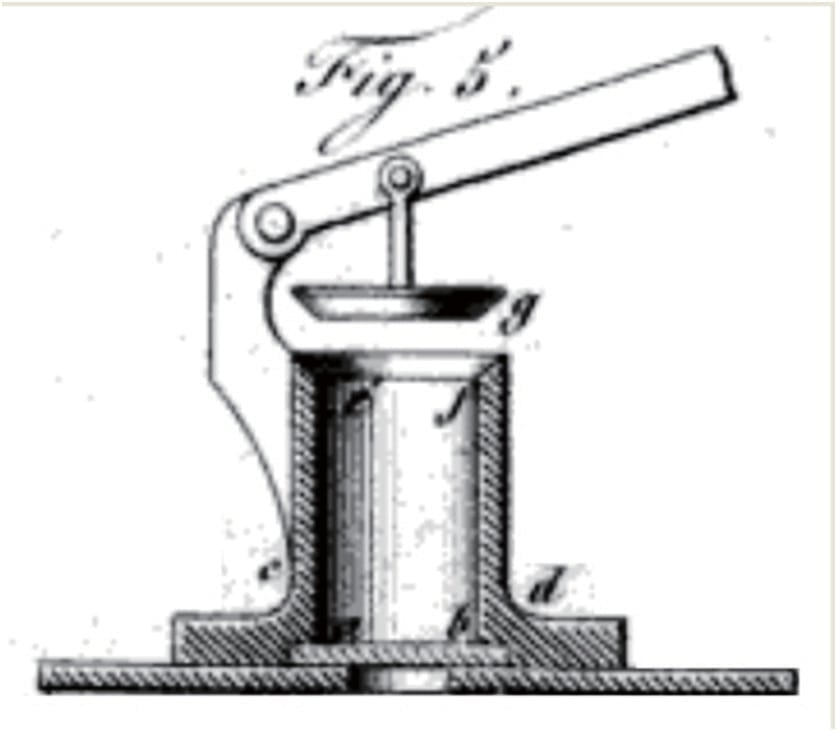
1890 De-multiplied fusible link assembled on a cable (Frederick Grinnel’s patent N°432403 )
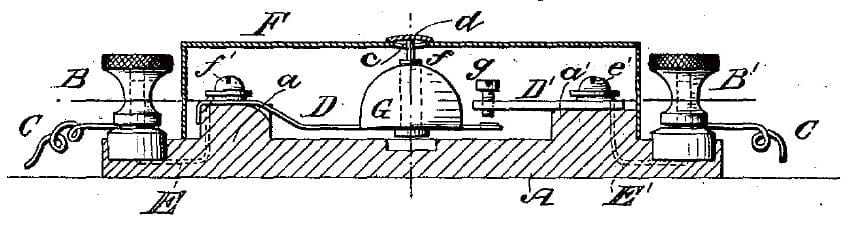
1884 Fire alarm closing an electrical contact using fusible alloy washer (d) (US Pat. Ross No. 298121)
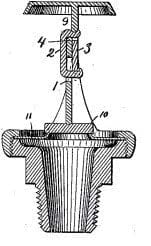
1890: Sprinkler head using parts welded together with a Wood’s fusible alloy and a leveraging effort mechanism (Frederick Grinnel’s patent N°432403 )
