Absolute pressure, barometric pressure,negative pressure, differential pressure
1. Definitions
Although pressure is an absolute value, the current measures pressure, are usually made relative to ambient air pressure. In other cases, the measurements are compared to vacuum. This leads to confusion and errors.
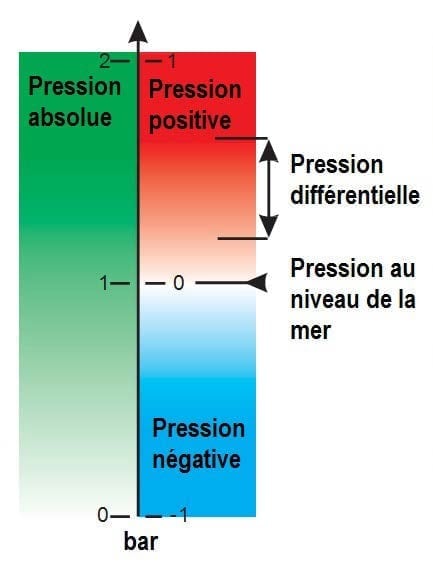
- The absolute pressure is the one that takes as its point zero the interstellar vacuum pressure. There is no negative pressure relative to this vacuum pressure
- Barometric pressure, also called relative pressure, which the zero point is taken at atmospheric pressure. The value, measured in absolute pressure of the atmospheric pressure is typically about 100 kPa at sea level, but varies with altitude and weather
- The negative-pressure is the pressure measured by regarding as zero the atmospheric pressure. It is expressed as the difference in pressure relative to atmospheric or barometric pressure and preceded by a minus sign.
The zero point reference used in pressure is often implied by the context, and is added only when clarification is needed. For example: The tire pressure and blood pressure are relative pressures by convention, while atmospheric pressure, the pressure of deep vacuum and altimeter pressure are absolute pressures.
The indication of moderate depression is often ambiguous, as with forgetfulness of the negative sign, it may represent an absolute pressure or abarometric pressure.
Thus, if the minus sign is forgotten, a vacuum of 25 kPa can be understood as an absolute pressure of 25 kPa (about 75 kPa below atmospheric pressure) or a barometric pressure of 25 kPa below atmospheric pressure
The differential-pressure is the pressure difference between two points. Differential pressures are commonly used in industrial processes. Differential pressure switchesand differential pressure gauges have two inlet ports, each connected to one of the volumes whose pressure is to be monitored.
2. Units
2.1 Legal Unit
Since 1971, the SI (International Sytem of Units) unit of pressure is the Pascal (Pa), equal to one newton per square meter (N/m²)
This unit name is entered into force in 1971, andbefore that, pressure in SI was expressed in N/m².
2.2 Traditional Units and notation
Sometimes the zero reference is shown in brackets after the unit, for example 101 kPa (abs).
The pound per square inch (PSI) is still widely used in the United States and Canada, specially for cars. A suffix is often placed behind PSI to indicate the zeroreference measurement : PSIA for absolute, PSIG for barometric pressure, PSID for differential pressure.
Because pressure was once commonly measured by its ability to displace a column of liquid in a graduated glass tube, pressures are often expressed as a length in mm or inches of a particular fluid such as mercury (Hg) or water.
Although dated, these units are still used in many areas: Blood pressure is measured in millimeters of mercury in most parts of the world, and respiratory pressures expressed in centimeters of water are still common. Natural gas pressures in pipeline are measured in inches of water, expressed as “WC” (“water column”.) In vacuum systems, the Torr (torr), the millimeters of mercury
(mmHg) and inches of mercury (inHg) are most commonly used.Torr and mmHg usually indicates an absolute pressure, while inHg usually indicates a relative pressure
Atmospheric pressures are usually expressed in kPa (kilopascals), or atmospheres (atm), except in meteorology where the hectopascal (hPa) and millibar (mbar) are preferred.
2.3 Other older units of pressure
- The barye (BA), was defined as equal to 1 dyn/cm² in the former CGS unit system
- The technical atmosphere (“at”) symbol) was defined as the pressure exerted by a column of water 10 meters: 1 “at”= 98066.5 Pa
- The normal atmosphere (atm) was defined in 1954 as being equal to 1,013,250 dynes per cm² (101 325 Pa). It corresponds to the pressure from a height of 760 mm of mercury at 0 ° C under normal acceleration of gravity (9.80665 m/s²). It represents the average atmospheric pressure at sea level using the latitude of Paris. It was used in technical documents during the period 1960-1980.
- The Torr (torr), was defined as being equal to 1/760 of an atmosphere, which was then represented by a column of mercury 760 mm
- Other hybrid units: mmHg/cm² or gram-force/cm², the kg / cm², he g/mol².
For the record, the use of the kilogram, gram, kilogram force or gram force (or their symbols) as a unit of force is prohibited in the SI system as the unit of force is the Newton (N).
