Corrosion resistance of aluminum
1. Corrosion resistance of aluminum
General corrosion resistance of aluminum enclosures when used outdoor
General information on the corrosion of aluminum
Aluminum and alloys generally have good resistance to atmospheric corrosion, in marine, urban and industrial environments. Molded under pressure, aluminum enclosures offer many possibilities of forms and treatments of the surface, require little maintenance and resist well in the time. In addition, with a view to sustainable development, aluminum is one of the metals whose recycling is the most economical. Aluminum remelting is only 5% of the energy required to make the metal from the ore. Aluminum naturally overlies with an oxide layer, which protects it most often against corrosion. In neutral aqueous solutions (4 <pH <9), this oxide film has a thickness of 50 Å and protects the metal (passivation). Aluminum is corroded homogeneously only in a very acidic solution, or in an alkaline solution. The strength and stability of the oxide layer depend on the ambient environment, the alloy composition and the microscopic structure of the metal (depending on the heat treatments applied). The electrochemical behavior of aluminum is influenced by the natural oxide film that governs the corrosion.
The boxes are massive and thick-walled (2 to 4mm); the corrosion is then superficial and affects only the appearance, without modifying the mechanical strength. In neutral environments, the overall corrosion rate of passive aluminum alloys is very small, but it is never totally zero. It thus keeps a value of the order of 5 μm per year, which can lead in the long run to a gradual change in the surface state to an unsightly appearance due to a change in roughness (orange peel).
The most common alteration is in the form of pitting corrosion which develops as cavities of varying depths. This is a very complex phenomenon, the mechanism of which is not fully determined.
In common industrial environments, aluminum housings can therefore be used without surface treatment other than deburring and standard vibration polishing.
1.2 Chemical composition
| Material and standards | Si | Cu | Mg | Zn | Mn | Fe | Ni | Sn | Ti | Al |
| EN AC 44300 DIN 1706 AlSi12(Fe) |
10.5 – 13.5 | <0.10 | – | <0.15 | <0.55 | <1 | – | – | <0.15 | remainder |
| ADC12 (JIS H5302:2000) | 9.6 – 12.0 | 1.5 – 3.5 | <0.3 | <1.0 | <0.5 | 0.6 – 0.9 | <0.5 | <0.2 | – | remainder |
2. Galvanic corrosion, also called Bimetallic Corrosion
Aluminum enclosures may be subject to a particular phenomenon which reduces their service life, can go as far as the perforation
of the envelope or the complete locking of the closure screws. It is galvanic or bimetallic corrosion.
Although most standards specify that appropriate safeguards must be in place to prevent galvanic corrosion on aluminum
enclosures, none advocates a solution or requires specific material or composition of alloys. However, even if the aluminum is
in an unfavorable position in the galvanic scale, it is most often covered with its passive film, which ennobles it considerably and
makes it much less sensitive to corrosion.
Galvanic (Bi-Metallic) corrosion is the additional corrosion that occurs when dissimilar metals are in contact in the presence of an
electrolyte (e.g. water, sea water). The corrosion of a metal, the anode, results from the positive current flowing from the anode
to the less reactive (more noble) metal, the cathode, through the electrolyte.
This process is similar to the conventional corrosion of a single, uncoupled metal but generally proceeds at a higher rate
depending on the difference in electrochemical reactivity of the anode and cathode metal.
There is a potential difference between the two metals that depends on both the metal and the solution. Two different metals or
two alloys in contact with the same medium usually take two different potentials. If these two metals are electrically connected,
their potential difference gives rise to electrochemical reactions and to the circulation of an electric current.
The most negative (the least noble) metal is positively polarized and the most positive metal is negatively polarized. In the vast
majority of cases, this configuration corresponds to an increase in the corrosion rate of the most corrodible metal (the most
negative), and a decrease in the corrosion rate of the least corrodible metal (the most positive).
Galvanic corrosion only occurs if the following 4 conditions are present and simultaneous:
- A: An electrolyte bridging the two metals.
When the conductivity of the electrolyte is low, corrosion is localized to the contact areas between the two metals. As the conductivity of the electrolyte increases, the corroded surface increases.
- B: An electrical contact between the two metals.
If the electrical contact is not established between the two metals by the interposition of an insulator (aluminum oxide,
phosphating, paint, oil, etc.), the current does not circulate, there is no corrosion.
- C: A difference in potential between the metals to enable a significant galvanic current .
The higher this value, the greater the electromotive force of the phenomenon. A difference of several hundred millivolts will result
in strong galvanic corrosion, whereas a difference of less than 200-300mV will not have significant consequences. These galvanic
corrosion potentials are given by a table which gives the electric potential of metals, usually measured by a so-called “Standard
Calomel Electrode (S.C.E.)” technique. (See below)
- D: A sustained cathodic reaction on the more noble of the two metals.
The ratio of the surfaces of the two metals
- The most unfavorable case is that of a large cathodic surface (the most positive material) electrically connected to a small anodic surface (the most negative metal). The corrosion rate of the most negative metal can be multiplied by 100 see per
For example, stainless steel screws enclosing an aluminum housing will be prone to corrosion due to surface differences.
Corrosion of the noble metal, and influence of the salts produced by its corrosion
- The corrosion resistance of the noblest metal, regardless of its potential, has a considerable influence on the behavior of the bimetallic corrosion. If the noblest metal corrodes, its corrosion products may, by displacement, accelerate the corrosion of the most corrodable metal. For example, copper, although considered as a noble metal and whose galvanic torque with aluminum is small, produces oxides that can corrode aluminum, which is a critical parameter when designing earth terminal blocks on aluminum housings that can accommodate copper
Sacrificial metal coatings
By applying to the cathodic side a sacrificial coating having a potential similar to or near that of the anodic member, the galvanic corrosion is reduced.
Main design rule:
- The sacrificial element should be on the anodic side and
- Be careful to use fasteners that have an intact Example:
Zinc plating on steel fasteners will sacrifice the zinc instead of corroding the Aluminum (Potential difference 100 to 200mV).
Caution:
Do not use nickel plated steel fasteners, as the potential difference (450mV) between aluminum and nickel is too high and will corrode aluminum.
Specific issue of galvanic corrosion between Stainless steel and aluminum
The corrosion potentials of the stainless steels are “cathodics” and located in the “noble” area and the corrosion potentials of aluminum are “Anodic” and located in the “non-noble” area, with a large potential difference. This means that there will be no galvanic corrosion on stainless steel when placed in contact with aluminum while aluminum will corrode.
Although aluminum is anodic to stainless steel, large relative surface areas of aluminum to stainless steel can be acceptable, dependent on local conditions.
Stainless steel fasteners in aluminum plates or sheets or massive parts are normally considered safe. Even with no insulation between the metals, there should be little risk of corrosion.
In contrast, in a marine environment, severe localized pitting corrosion to the aluminum treads has been observed where un- insulated stainless steel screws were used.
Mechanical methods of reducing galvanic corrosion between aluminum and stainless steel
- Isolating the two materials by means of an electrical insulating material, like plastic, wherever
- Avoid relatively small areas of the less noble metal (Aluminum) and large areas of the more noble metal (Stainless steel).
NB: Coupling a relatively wide area of aluminum with a small surface area of a cracked stainless steel part can cause a rapid attack of the material inside the crevice and corrode the stainless steel.
- Protect against the electrolyte presence around the bimetallic junction. For example, if possible, paint both
- Apply corrosion inhibitors under screw heads and threads
- Apply an insulating organic coating to the contact surfaces before
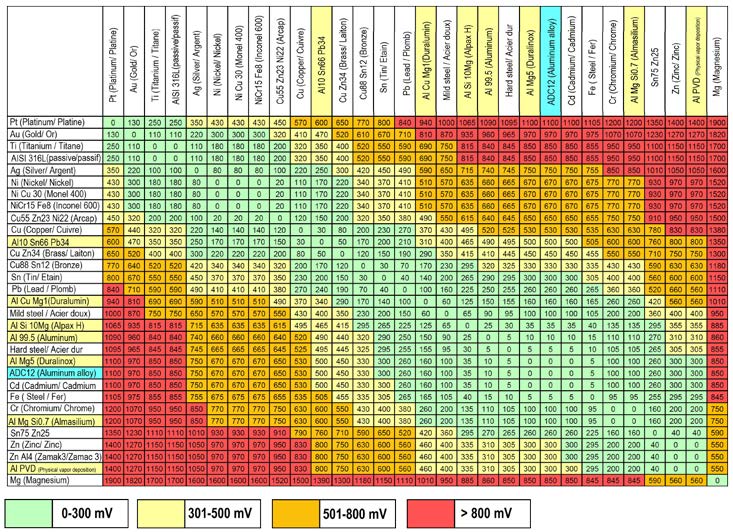
Electrochemical couples between aluminum alloys
Table of electrochemical voltage between aluminum alloys (Names highlighted in yellow and blue) and other common metals, in a 2% salt water solution.
There is no noticeable occurrence of corrosion when the value of the galvanic torque is less than 300mV.
4.1 Other limitations in the use of aluminum alloys
To avoid ignition hazards due to impact or friction, standards for explosion-proof equipment (IEC 60079-0) restrict the use of aluminum.
Enclosures for group I:
The total weight of aluminum + magnesium + titanium should not exceed 15% of the total weight (or 6% of the total weight of magnesium + titanium alloys having no aluminum), which eliminates, for that group, most alloys containing aluminum.
Enclosures for Group II:
– For zone 0: the total weight of aluminum + magnesium + titanium + zirconium should not exceed 10% of the total weight (or 7.5% of the total weight of magnesium + titanium + zirconium alloys for not containing aluminum ), which eliminates, for this group and this zone most alloys containing aluminum
For zone 1: the weight of magnesium should not exceed 7.5% of the total weight
For zone 2: no specifications.
